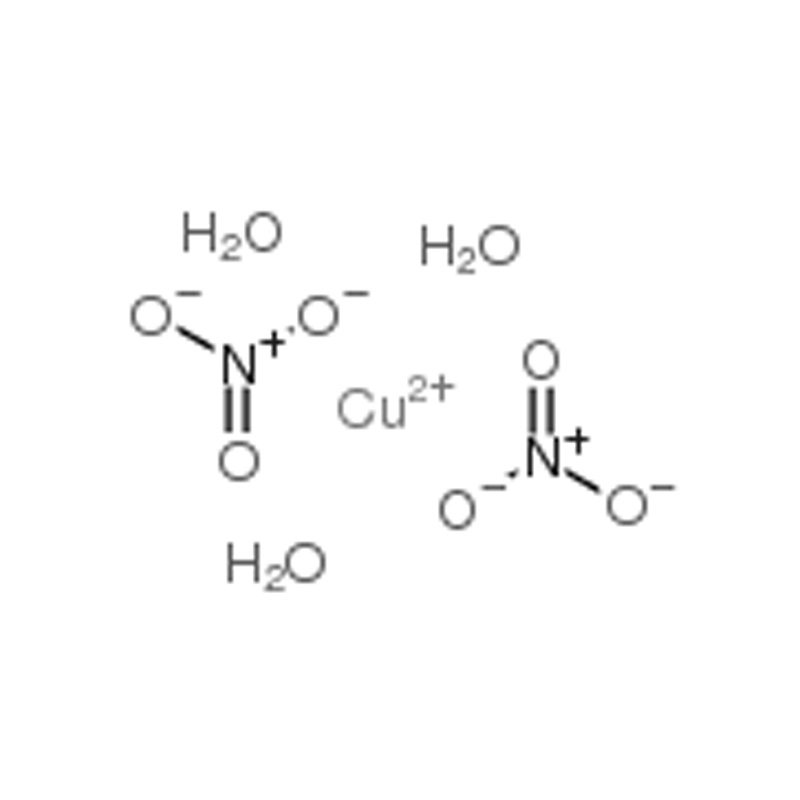
Cupric Nitrate Trihydrate CAS# 10031-43-3
Cupric nitrate is the copper salt of nitrate and has a wide range of applications. A primary use is its conversion to copper oxide, a powerful catalyst in organic chemistry. It is also employed in textiles, metal polishing, and can be used to demonstrate voltaic cell reactions in educational settings.
In organic synthesis, cupric nitrate can react with acetic anhydride as an effective reagent for the nitration of aromatic compounds. Its hydrate form can catalyze the oxidative coupling of 2,6-dimethylphenol to produce polymers used in engineering materials, while the trihydrate form can catalyze the solvent-free synthesis of 3-aminopropenones and 3-aminopropenoates. Additionally, the pentahydrate form is useful in preparing novel coordination polymers.
Cupric nitrate also finds applications in medicine, insecticides, chemical analysis, and the production of light-sensitive papers, highlighting its versatility across industries.
![Cupric nitrate trihydrate CAS#10031-43-3 Cupric nitrate trihydrate CAS#10031-43-3]()
Cupric nitrate trihydrate Chemical Properties
| Melting point | 114 °C |
| Boiling point | 170°C |
| bulk density | 1050kg/m3 |
| density | 2,32 g/cm3 |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 2670g/l |
| form | Fine Powder |
| Specific Gravity | 2.32 |
| color | Cream to brown |
| PH | 3-4 (50g/l, H2O, 20℃) |
| Water Solubility | soluble |
| Stability: | Stable. Strong oxidant - may ignite combustible material. Moisture sensitive. Incompatible with anhydrides, ammonia, amides, cyanides. |
| InChI | InChI=1S/Cu.HNO3.H2O/c;2-1(3)4;/h;(H,2,3,4);1H2 |
| InChIKey | VVHXYVGAMODMHK-UHFFFAOYSA-N |
| SMILES | N(O)(=O)=O.[Cu].O |
| CAS DataBase Reference | 10031-43-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Copper dinitrate(10031-43-3) |
Safety Information
| Hazard Codes | O,C,Xn,N |
| Risk Statements | 8-22-34-36/38-50/53-41-38 |
| Safety Statements | 17-26-36/37/39-45-37/39-61-60-39 |
| RIDADR | UN 3085 5.1/PG 2 |
| WGK Germany | 2 |
| RTECS | GL7875000 |
| F | 3 |
| HazardClass | 5.1 |
| PackingGroup | II |
| HS Code | 28342930 |
| Storage Class | 5.1B - Oxidizing hazardous materials |
| Hazard Classifications | Aquatic Acute 1 |
| Aquatic Chronic 2 |
| Eye Dam. 1 |
| Ox. Sol. 2 |
| Skin Corr. 1B |
| Toxicity | LD50 orally in Rabbit: 940 mg/kg |
![Cupric nitrate trihydrate CAS#10031-43-3 Cupric nitrate trihydrate CAS#10031-43-3]()
Product Application of Cupric Nitrate Trihydrate CAS# 10031-43-3
Copper(II) nitrate trihydrate can be used in the synthesis of various mononuclear copper(II) complexes, including complexes formed with ligands such as 1,10-phenanthroline (phen), dipyrido[3,2-d:2′,3′-f]quinoxaline (dpq), and dipyrido[3,2-a:2′,3′-c]phenazine (dppz). These complexes are commonly studied and applied in coordination chemistry and related research fields.
![Cupric nitrate trihydrate CAS#10031-43-3 Cupric nitrate trihydrate CAS#10031-43-3]()